Acid Dyes
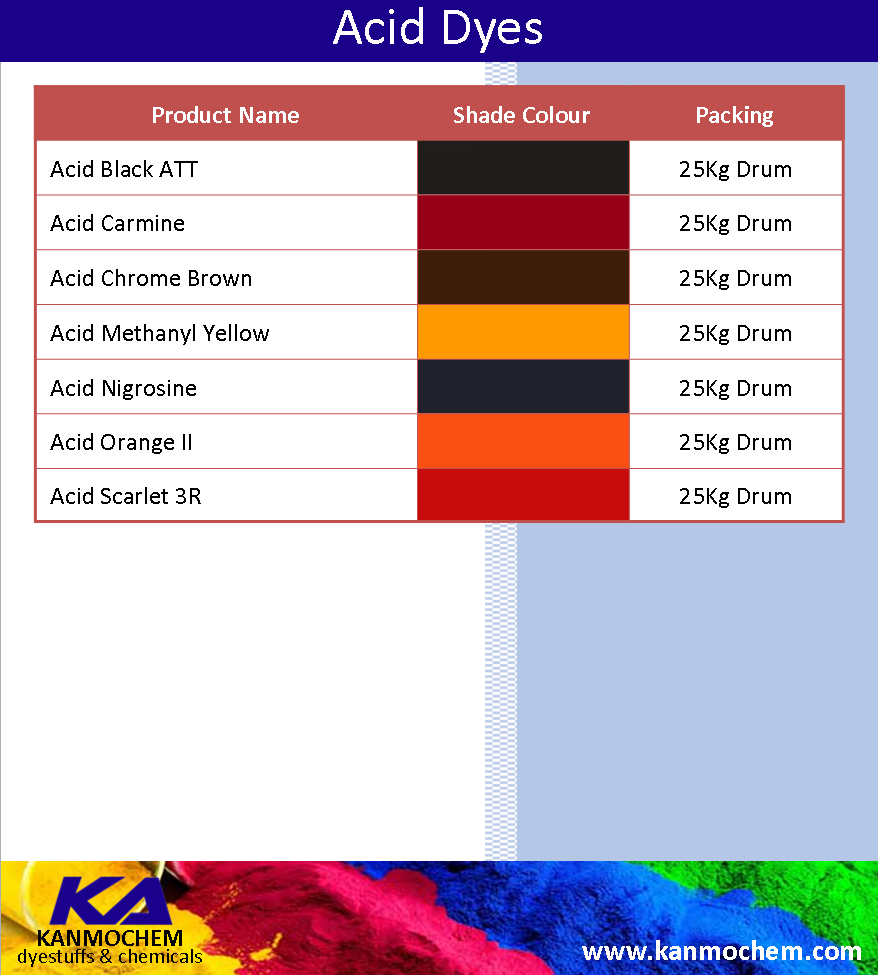
Kanmochem Dyestuffs & Chemicals has a variety of quality acid dyes ranging from Acid Black ATT, Acid Carmine, Acid Chrome Brown, Acid Methanyl Yellow, Acid Orange II and Acid Scarlet 3R.
Properties of Acid Dyes
Acid dyes are highly water soluble, and have better light fastness than basic dyes. The textile acid dyes are effective for protein fibers such as silk, wool, nylon and modified acrylics. They contain sulphonic acid groups, which are usually present as sodium sulphonate salts. These increase solubility in water, and give the dye molecules a negative charge. In an acidic solution, the -NH2 functionalities of the fibres are protonated to give a positive charge: -NH3+. This charge interacts with the negative dye charge, allowing the formation of ionic interactions. As well as this, Van-der-Waals bonds, dipolar bonds and hydrogen bonds are formed between dye and fibre. As a group, acid dyes can be divided into two sub-groups: acid-leveling or acid-milling.
Product Catalogue